prepFAST For USP <232> & <233>

Automated Elemental Analysis for Pharmaceutical Laboratories
![]() The prepFAST inline autodilution system with ICP and ICP-MS, fully automates USP <233> methods. The prepFAST can:
The prepFAST inline autodilution system with ICP and ICP-MS, fully automates USP <233> methods. The prepFAST can:
- dilute a single stock standard to build linear calibration curves;
- dilute samples to the appropriate TDS;
- spikes samples at user defined J values.
The USP Chapter <232> defines a target (J) value as a function of PDE (μg/day) and a drugs daily dose (g/day), whereas Chapter <233> outlines specific protocols for the determination of toxic metals by ICP/ICP-MS. USP <233> requires a calibration curve and a series of QC validation protocols including spike recovery, accuracy, precision and stability tests to be based on the target values (J).
To learn more on USP <232> and <233>, please contact us by filling out the online form.
Autocalibration
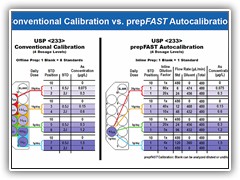 Automatically calibrate for multiple products from the same standard
Automatically calibrate for multiple products from the same standard
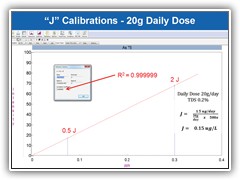 Ensure linear calibrations by reducing contamination & eliminating manual dilution error
Ensure linear calibrations by reducing contamination & eliminating manual dilution error
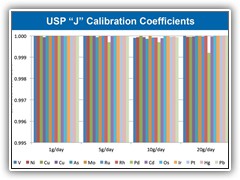 Easily calibrate for all elements in USP 232
Easily calibrate for all elements in USP 232
Automated Sample Dilution
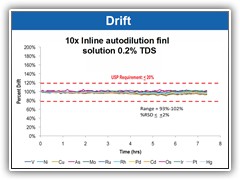 Inline sample dilution ensures minimal drift, allowing for uninterrupted sample analysis
Inline sample dilution ensures minimal drift, allowing for uninterrupted sample analysis
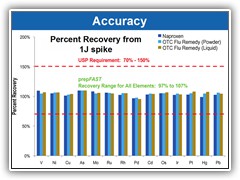 Surpasses all USP validation criteria
Surpasses all USP validation criteria
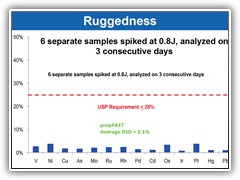 Surpasses all USP validation criteria
Surpasses all USP validation criteria
Auto “J” Spiking
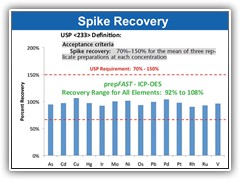 Automate QC and Validation spiking to completely remove sample preparation
Automate QC and Validation spiking to completely remove sample preparation